CLINICAL SAFETY MANAGEMENT IN ANZ
Structured clinical safety built into the Dedalus product lifecycle
Structured clinical safety built into the Dedalus product lifecycle
Dedalus operates a global Clinical Safety Management System (CSMS), with regional implementation across Australia and New Zealand, embedded within our global Quality, Regulatory and Assurance (QARA) organisation. This integrated approach ensures that clinical safety, regulatory compliance, and product delivery remain aligned across the Dedalus suite of products.
In addition to operating a structured Clinical Safety Management System aligned with international standards, our regional Clinical Safety Officers have contributed as subject matter experts (SMEs) to the review and development of the ADHA/AIDH Digital Health Clinical Safety courses. This involvement reflects our clinical safety governance expertise and our active engagement in strengthening national digital health safety capability.

Aligned with recognised international standards
Dedalus’ clinical safety approach is ISO9001 certified and aligns with established standards:
- DCB 0129 – Clinical Risk Management in the manufacture of health IT
- DCB 0160 – Clinical Risk Management in the deployment and use of health IT
These frameworks are widely regarded as leading international benchmarks for digital health safety and underpin emerging regulatory expectations globally.
Across the Dedalus product suite, this approach includes:
- Formal clinical risk assessments
- Hazard identification and mitigation
- Development and maintenance of clinical safety cases
- Ongoing monitoring and post-deployment review
- Clear clinical safety governance
Australia is moving toward more formal clinical governance expectations for digital health, as reflected in national frameworks and performance reporting from the Australian Digital Health Agency.
Because Dedalus already operates a structured CSMS:
- Clinical risk management processes are established across the product lifecycle.
- Safety cases are developed and maintained where applicable.
- Clinical safety oversight is embedded within the regional QARA function.
This places Dedalus in a strong position to align with emerging national requirements as they evolve.
Integrated clinical safety and regulatory oversight
Within the ANZ region, Clinical Safety Officers operate as part of the QARA team, ensuring alignment between:
- Clinical safety activities
- Product classification and regulatory strategy
- ARTG and WAND notifications
- Post-market monitoring and reporting
- Advertising and promotional material compliance
This integrated structure ensures that clinical safety, regulatory obligations, and product delivery are managed cohesively.
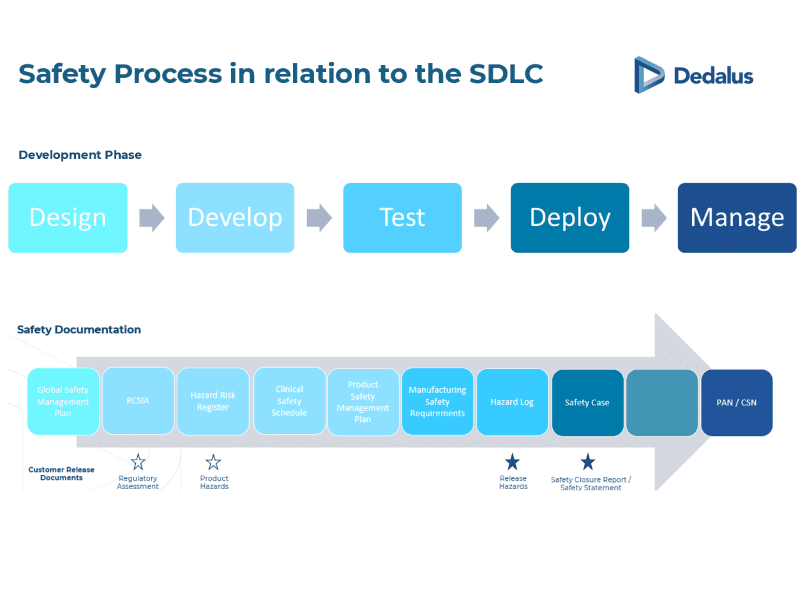
Lifecycle clinical safety approach
For relevant functionality within the Dedalus product suite, the CSMS follows a lifecycle model:
- Clinical risk assessment
- Safety case development
- Implementation and validation
- Post-deployment monitoring
- Safety case updates with each software change
This iterative process supports continuous risk management and ongoing safety assurance, ensuring that risks are proactively managed and reduced to as low as reasonably practical (ALARP).
Benefits for healthcare organisations
- Structured clinical safety aligned with international best practice
- Documented safety cases where applicable
- Transparent risk assessment matrices and risk acceptance criteria
- Notification of serious incidents
- Integrated regulatory and clinical oversight
- Alignment with emerging Australian digital health governance expectations
- A mature, globally supported clinical safety capability
What is included in a safety case & how can my organisation use it?
The safety case provides documented assurance that clinical risks associated with the product have been systematically identified, assessed, and managed.
It includes:
- The results of the hazard assessment, including a summary of safety-related scope items, associated hazards, and evidence of mitigation undertaken by Dedalus to minimise clinical risk to as low as reasonably practical (ALARP)
- A summary of open safety-related items recorded in the incident management system
- A Regulatory and Contractual Safety Impact Assessment (RCSIA), outlining any regulatory or contractual considerations linked to the identified risks.
Customers are encouraged to review the Safety Case within the context of their business processes, to inform their own local clinical governance processes, validate implementation controls and ensure appropriate mitigations are embedded within operational workflows.